Main navigation
Bacteria produce bioplastics: resource-saving and very environmentally friendly
Using living cells as mini-factories to produce plastic from nothing more than water, sunlight and carbon dioxide; plastic that is also 100 percent degradable – it sounds far-fetched but it actually works: researchers at the University of Tübingen have genetically engineered cyanobacteria so that they fill their cells to the brim with polyhydroxybutyrate. The researchers are now turning the idea into reality with the development of pilot plants that can produce this bioplastic on a larger scale.
It is impossible to imagine our everyday lives without plastic; it is used for producing packaging, textiles, adhesives and car parts. However, producing synthetic plastics requires fossil resources, which are finite. Moreover, these plastic products are not biodegradable and thus pollute the environment, as the majority of them accumulate in nature or in landfills instead of decomposing.1) Plastic pollution is therefore becoming an increasing problem: plastic waste is already ubiquitous in all large ocean basins as well as in many freshwater areas and land habitats.
This is why researchers around the world have been working for quite some years at full stretch to come up with more sustainable alternatives for producing plastic. Although technologies to produce bioplastics from various natural sources such as starch, cellulose or lactic acid are available, some even on an industrial scale, they only make up a very small proportion of the total amount of plastic produced. In 2021, bioplastic products only amounted to about 6 percent.2) Moreover, although they are much more environmentally friendly than their petrochemical counterparts, bioplastics are not always 100 percent biodegradable. PLA (polylactate), which is currently the most commonly used bioplastic, does not decompose in salt water. The solutions that have been found so far are therefore still far from satisfactory.
Cyanobacteria: plastic producers that do not use arable land

Polyhydroxyalkanoates (PHAs) represent a promising option. They are made from natural raw materials and also degrade much better than their petrochemical counterparts. The most abundant PHA is polyhydroxybutyrate (PHB), which forms in the natural metabolism of various microorganisms almost on its own. PHB has similar material properties to polypropylene, a commonly used conventional plastic, and is malleable when heated. PHB production using heterotrophic bacteria is therefore already a well-established method, even in larger production plants. However, these microbial plastic producers need energy and must be given food in the form of sugar, thus tying up resources that are needed for human nutrition.
On the contrary, phototrophic organisms that only need light as an energy source and thus synthesise PHB in a completely carbon neutral way do not need arable land to grow. Cyanobacteria are representatives of this group of organisms. "The metabolism of these ecologically relevant microorganisms can be easily manipulated biotechnologically," explains Prof. Dr. Karl Forchhammer, who heads the Department of Microbiology/Organismic Interactions at the University of Tübingen and has been studying cyanobacteria for three decades. "We’ve known for a long time that cyanobacteria are natural PHB producers. However, initially we were mainly interested in finding out why they produce PHB at all. Once we had understood why, we started to focus our research more on application. That was around two years ago."
Cells bulging with fully degradable plastic
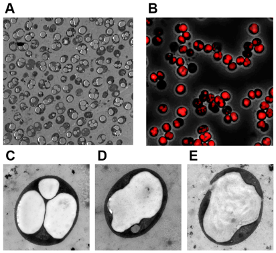
The researchers assume that cyanobacteria use lipid-like PHB as a kind of storage material. However, this only occurs in quantities of about 15 percent per cell dry weight (CDW), quantities that are too low to be anywhere near financially viable. This is why the researchers from Tübingen worked on increasing PHB synthesis rates.
And they were successful! Forchhammer, together with his former doctoral student Dr. Moritz Koch, created a new cyanobacterium strain that contains up to 81 percent PHB per CDW.3) The polymer produced consists of pure PHB, which is highly isotactic, meaning that the side groups of this macromolecule are arranged in a regular pattern. This is a prerequisite for the key properties required for plastic. "I would never have believed it possible," Forchhammer says. "These quantities are the highest ever achieved in a known cyanobacterium and the best basis for producing a sustainable product in a sustainable way." And not just in terms of feedstock! The bioplastic produced by cyanobacteria can be degraded completely within six months. "Everything nature creates, it can also degrade. And this is a huge advantage," the expert adds.
Info box: Cyanobacteria – Phylogenetically ancient organisms for modern applications
Were it not for their tiny size, cyanobacteria could be called living dinosaurs because they are among the oldest known life forms on earth. They colonised our planet around 3 billion years ago and played a key role in the transition to the Earth's oxygenated atmosphere. This is because unlike other prokaryotes, cyanobacteria are capable of photosynthesis. This is why these green microorganisms were for a long time called 'blue-green algae' rather than being counted as 'real' bacteria. The term 'blue-green algae' is misleading as they are not eukaryotes, and hence not algae. Due to their ability to produce oxygen, cyanobacteria are still hugely important for our environment today. They are found in practically all ecosystems on earth - including and notably in extreme habitats: in fresh and salt water, on stones, in the ground, in hot springs and even in Antarctic ice. However, the mass colonisation of a body of water with cyanobacteria can also become a problem.
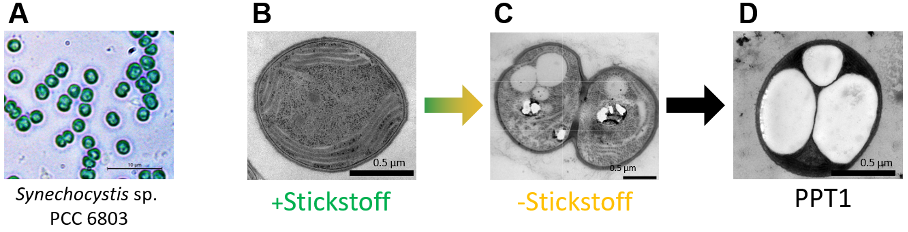
The freshwater cyanobacterium Synechocystis sp. PCC 6803, the species also used at the University of Tübingen, is a well-studied model organism that can be easily cultivated in the laboratory and in bioreactors. Biotechnological modifications can be used to specifically intervene in the bacterial metabolism for various applications.
Plastic production actively contributes to CO2 reduction
Photosynthesis is the first step in the production of the sustainable bioplastic. Cyanobacteria carry out biosynthesis to synthesise cell building blocks from CO2, sunlight and water. This is in principle the same thing plants do. However, cyanobacteria switch to survival mode when their growth is restricted in some way or other: instead of producing essential cell building blocks, they now produce glycogen, a small amount of which is subsequently converted into PHB.
The biologists from Tübingen made a decisive discovery related to just this: they discovered the switch that controls the distribution of fixed carbon in the cell. As soon as this switch is removed and the cells are also deprived of nitrogen, the fixed carbon is no longer turned into glycogen, but into PHB. PHB can easily be extracted from the cells with the help of solvents and used as a raw material for practical applications.
But that's not all: since photosynthesis consumes CO2 in not inconsiderable quantities, the production of bioplastic in this case not only conserves resources, but can also actively contribute to the reduction of this greenhouse gas. The project will study the concrete use of the process as a CO2 trap in combination with CO2 production plants. "This additional benefit is what makes the process profitable from the start," says Forchhammer. The researchers were awarded second place in the Science2Start ideas competition in late 2021 for their sustainable bioplastic.
Upscaling offers even more potential for sustainability
The next step will be to transfer the process from the laboratory to the industrial scale. "This will once again require a lot of new technology," explains Forchhammer, who of course continues to accompany this process from a bioscientific perspective. "I am not a plant manufacturer. And in fact, we had not anticipated being able to get this far by now. But our second place in the competition led to a lot of interest to the extent that we decided to look at working collaboratively with others in an interdisciplinary team to be able to start transferring the process to the industrial scale."
The first upscaling step will involve the researchers studying the cultivation of the living mini-factories in 500-l tanks. Productivity will subsequently be increased on a modular basis, possibly also using flatbed reactors. Many parameters have to be taken into account, such as the right light dosage and water quality. The kind of water used has particular potential in terms of achieving even greater sustainability: "Perhaps it will also be possible to use waste water or even salt water. These would be incredibly cheap resources," says the biologist.
The researchers also intend to utilise the biomass that remains after PHB has been extracted. One idea is to extract pigments. Of course, it would also theoretically be possible to use the cyanobacteria to produce completely different raw materials. "Intensive work is being done worldwide on the development of such green cell factories, which could be used to produce a broad range of things, from biofuel to ethylene for the chemical industry. We are not the only ones," says Forchhammer. "But we are definitely at the forefront as far as bioplastics are concerned. Although we do not yet have the definitive answer, we have already come up with a well-functioning prototype."
References:
1) Geyer, R. et al. (2017): "Production, use, and fate of all plastics ever made". Science Advances 3, 7: www.science.org/doi/10.1126/sciadv.1700782.
2) Institute for Bioplastics and Biocomposites IfBB: "Biopolymers – facts and statistics 2021". www.ifbb-hannover.de/files/IfBB/downloads/faltblaetter_broschueren/f+s/Biopolymers-Facts-Statistics-einseitig-2021.pdf
3) Koch, M. et al. (2020): "Maximizing PHB content in Synechocystis sp. PCC 6803: an new metabolic engineering strategy based on the regulator PirC." Microb Cell Fact.19(1):231. https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-020-01491-1.